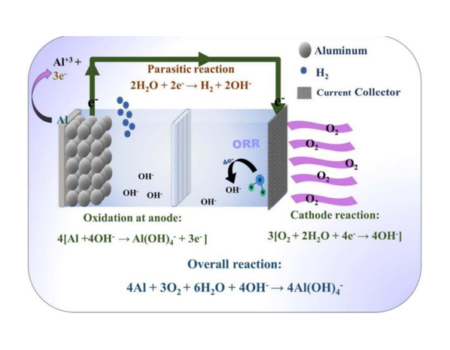
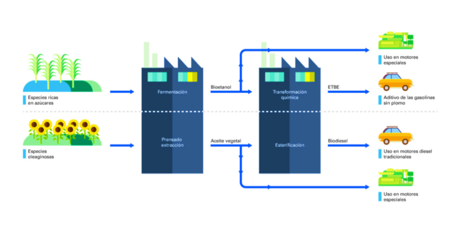
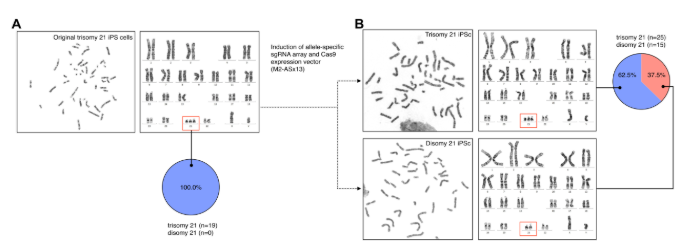
Full Time Professor
Bioengineering and Chemical Engineering Department
The holine and endolysine proteins are responsible for lysis during infection of the host bacteria by the bacteriophage. Endolysine performs cell wall degradation and is a type of muriatic enzyme, which accumulate in the cytosol during viral infection. On the other hand, holins are low molecular weight membrane proteins that accumulate in the folded form of the membrane. Subsequently, a “programmed” mechanism in the holine gene makes the membrane permeable so that fully folded endolysine can trigger wall degradation. Endolysine destroys murein in a micro-explosion effect. The importance of holins lies in that they control the infective cycle by signalling the activation of lytic phages. The efficiency of infection depends on the optimal timing of this signal. To date, more than 30 groups of holins have been reported. Many holin inhibitors have been described, so part of their initiation and negative control of activity is known [1].


Endolysins and holins are enzymes that bacteriophages use during their lithic process against their hosts, which has led to a growing interest in their application against multidrug-resistant bacteria such as Salmonella infantis as an alternative to antibiotics. Although they have remarkable antibacterial activity, their expression and production in bacterial systems present challenges that must be solved to ensure efficient production. Holins, when expressed recombiningly, tend to have low solubility due to their membrane protein nature, resulting in the formation of inclusion bodies that hinder their proper folding and purification [2]. Despite overcoming these limitations, both enzymes require a complicated cell breakdown and purification process for their application [2]. In this context, the M13 phage could be an effective tool not only to combat MDR bacteria but also to facilitate delivery of endolysins and holins. The synergy between M13 and these enzymes could optimize its antibacterial action and simplify its production. Cui et al. [4] used this technique to express holins of a Pseudomonas aeruginosa bacteriophage in the capsid of M13. Their results showed a significant growth inhibition halo of Pseudomonas aeruginosa compared to the use of holine unexpressed in the bacteriophage capsid. The phage delivery system with M13 bacteriophage, also used to express toxic proteins such as Gef and ChpBK, resulted in a significant reduction of bacteria in the blood of mice infected with E. coli. The CFU assay showed a progressive decrease in viable bacteria, with reductions of up to 948 times for Gef and 1579 times for ChpBK [5]
In another study submitted [6], it was found that phage M13 and its protein g3p can occlude the conjugative pilus, essential for gene transfer between bacteria. By preventing the formation of conjugative pairs, M13 phage reduces conjugation efficiency, limiting the spread of antibiotic resistance genes. Unlike traditional antibiotics, which may be ineffective against multidrug-resistant bacteria due to their resistance mechanisms, phages like M13 have the ability to introduce genes that encode lethal proteins directly into bacteria. This can cause cell death through mechanisms other than antibiotics, allowing the pathways of bacterial resistance to be bypassed.
References
[1] Wang IN, Smith DL, Young R. Holins: the protein clocks of bacteriophage infections. Annu Rev Microbiol. 2000;54:799-825. doi: 10.1146/annurev.micro.54.1.799. PMID: 11018145.
[2]Ž. Alaksandr et al., “Efficient matrix-assisted refolding of the recombinant anti-staphylococcal truncated endolysin LysKCA and its structural and enzymatic description,” Protein Expr Purif, vol. 174, p. 105683, Oct. 2020, doi: 10.1016/J.PEP.2020.105683.
[3] M. T. P. Gontijo, G. P. Jorge, and M. Brocchi, “Current Status of Endolysin-Based Treatments against Gram-Negative Bacteria,” Antibiotics 2021, Vol. 10, Page 1143, vol. 10, no. 10, p. 1143, Sep. 2021, doi: 10.3390/ANTIBIOTICS10101143.
[4] J. Cui et al., “Characterization of a lytic Pseudomonas aeruginosa phage vB_PaeP_ASP23 and functional analysis of its lysin LysASP and holin HolASP,” Front Microbiol, vol. 14, p. 1093668, Mar. 2023, doi: 10.3389/FMICB.2023.1093668/BIBTEX.
[5] Westwater, Caroline et al. “Use of genetically engineered phage to deliver antimicrobial agents to bacteria: an alternative therapy for treatment of bacterial infections.” Antimicrobial agents and chemotherapy vol. 47,4 (2003): 1301-7. doi:10.1128/AAC.47.4.1301-1307.2003
[6] Lin, Abraham et al. “Inhibition of bacterial conjugation by phage M13 and its protein g3p: quantitative analysis and model.” PloS one vol. 6,5 (2011): e19991. doi:10.1371/journal.pone.0019991
[7]A review on re-emerging bacteriophage therapy in the era of XDR . POOJA BHADORIYA1; RICHA SHARMA1; REKHA MEHROTRA1; SIMRAN KAUR1; ISHA SRIVASTAVA1; MUKUL JAIN2; PRASHANT KAUSHIK3 BIOCELL Tech Science Press, 2023 47(9): 1915-1930